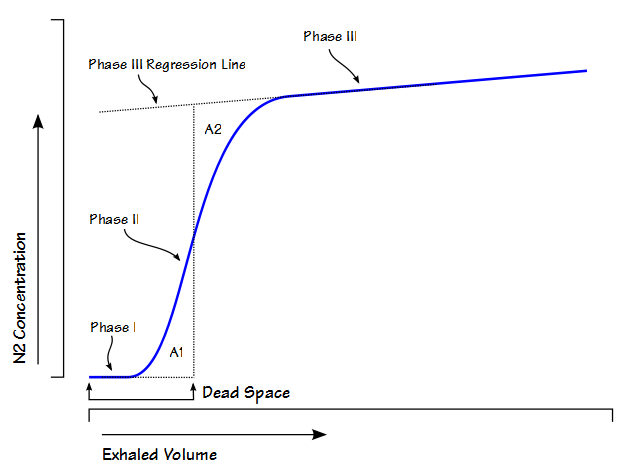
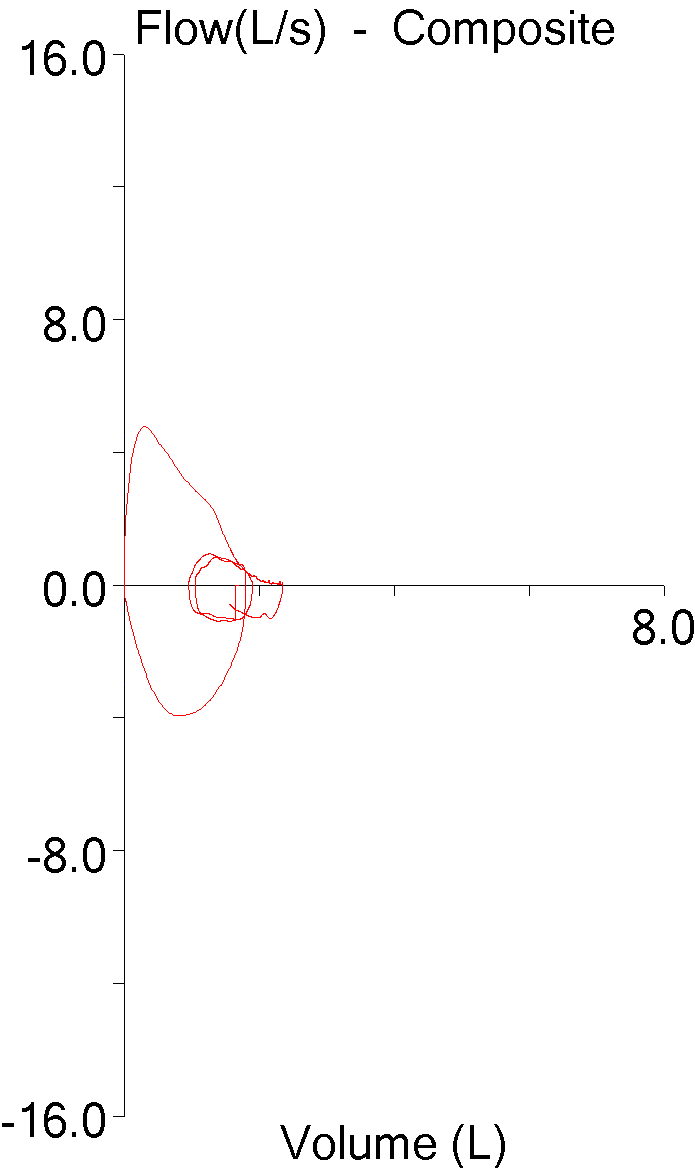
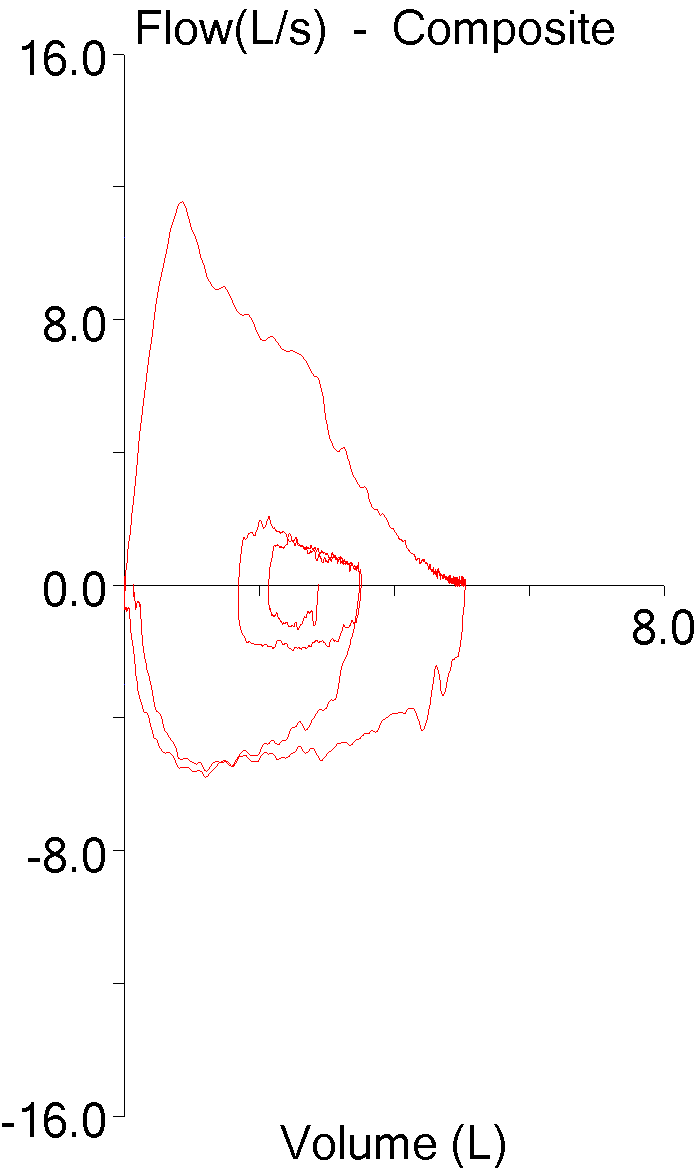
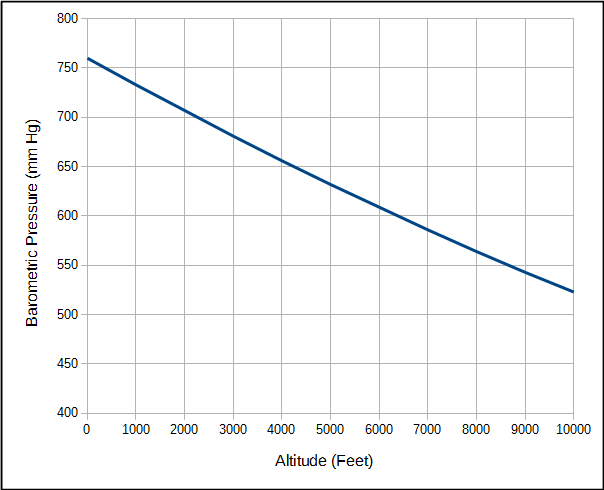
My lab is in the final stages of a software update that will allow for electronic signing of our reports. This has been a long and slow process partly because the release date of the software got pushed back several times but mostly because a wide variety of different hospital departments and sub-departments have had to be involved.
In all the years that I’ve had computers in the pulmonary function lab I’ve never gone through a software update that was either as easy as expected or occurred within the original schedule. This includes the time when all we had was a single IBM PC/AT with a 40 megabyte hard drive, no network and the only people that cared we were going through an update was ourselves. Since we now have a dozen networked PCs located in two different building on-campus as well as three off-site locations using an IS-managed SQL server and HL7 interface I didn’t have any expectations for a speedy update and so far I have not been disappointed.
This time because the update revolves around electronic signing the hospital’s Health Information Management (HIM, i.e. Medical Records) department has been significantly involved. Among other things this has led to HIM reviewing all of our reports and requiring changes to bring them up to hospital standards. To some extent this make sense since, for example, they require that patient identification be exactly the same on all reports from all departments (same fields, same locations).
However, they also questioned some of the terminology used on our test reports. We’ve used the default test names that were in our report format editor (yes, we’re that lazy) and until they were brought to our attention I never really thought how odd some of them were. In particular, some of the terms used for the diffusing capacity didn’t make a lot of sense. For example, DLCO corrected for hemoglobin was DsbHb and DLCO/VA was reported as D/Vasbhb. To some extent I understand where these names came from but the reality is that they are in part holdovers from the past, in part they come from a need to keep names short so they fit in what space is usually available on reports, and in some cases they were probably created by programmers who hadn’t the slightest idea what the correct nomenclature should have been.
Note: Dsb likely comes from a time when you needed to differentiate between the results of different types of DLCO tests (steady-state and single-breath). Since there hasn’t been a test system built for at least 40 years that could perform a steady-state DLCO, the need to make this distinction is long since past.
Continue reading →