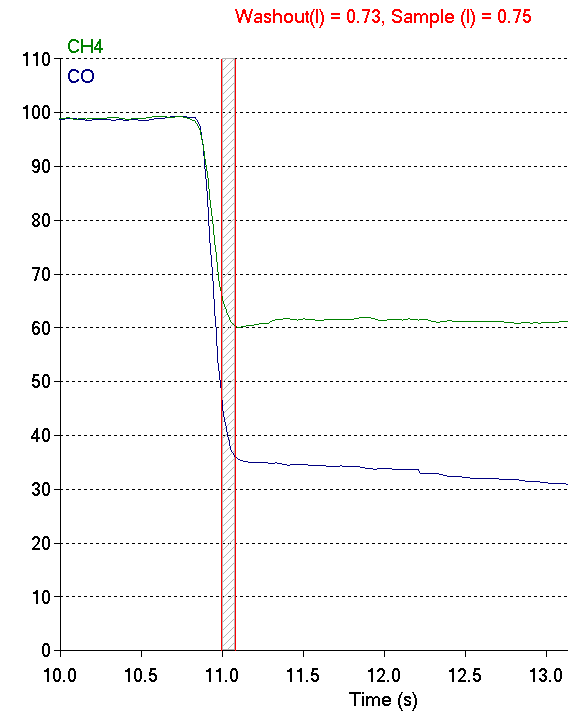
Recently while reviewing PFT reports I ran across a test from a patient who had been having spirometry, lung volume and DLCO tests performed at regular intervals for the last several years. Compared to the last several set of tests the most recent DLCO had decreased significantly while the FVC, FEV1 and TLC hadn’t changed. I took a closer look at the raw data from the DLCO test and when I did I saw that the washout volume was not correct.
Or more correctly, even though the washout volume matched the ATS/ERS standard for DLCO testing it was evident the expiratory gas sample was not taken from the alveolar plateau. The CO and CH4 concentrations at this point in the exhalation are higher than they are in the alveolar plateau and this means the reported DLCO was underestimated.
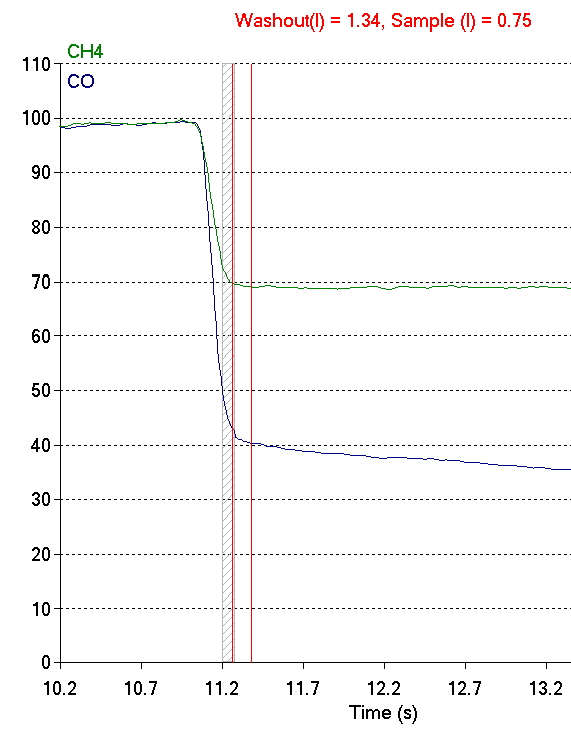
When I re-adjusted the washout so the gas sample was taken from the alveolar plateau, the DLCO went from 18.56 ml/min/mmHg to 22.26 ml/min/mmHg, which is a 20% increase and far more in line with the patient’s prior DLCO test results.
This, however, increased the washout volume from 0.75 L to 1.34 L. Why was the washout volume so high? The answer is it probably wasn’t.