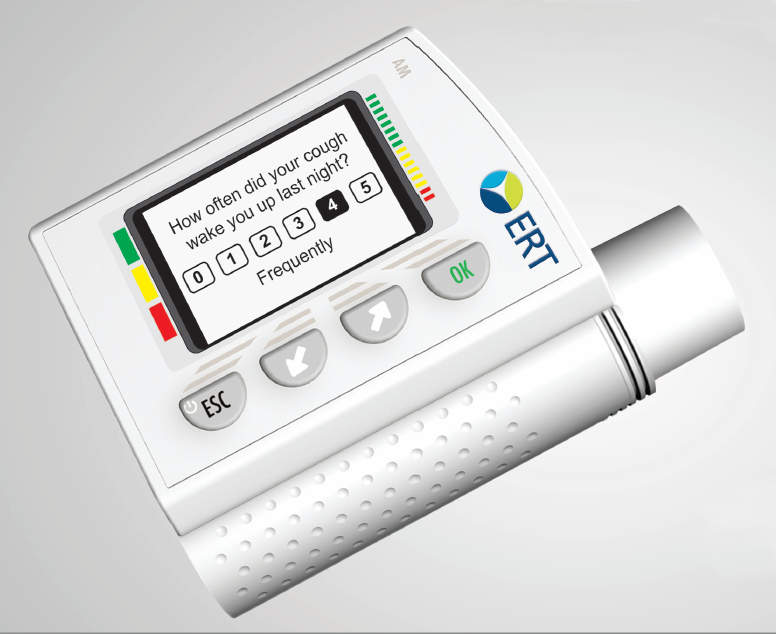
| Product Type |
: |
Spirometer |
| Product Name |
: |
AM3 |
| Company Name |
: |
ERT |
| |
|
|
| Intended for |
: |
Personal, Clinical Trials |
| Configuration |
: |
Hand-held, downloads to PC via RS232, USB. Bluetooth or Cellular GSM optional. |
| Sensor Technology |
: |
Turbine |
| Vol Range & Accuracy |
: |
0.5-8 Liters |
| Flow Range & Accuracy |
: |
0 to 14 L/sec |
| Calibration |
: |
Not specified |
| Power Requirements |
: |
Rechargable Li – Ion |
| TESTS: |
|
|
| FVC |
: |
FEV1 and Pef |
| Pre/Post |
: |
No |
| SVC |
: |
No |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
LCD 255×160 pixel |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
No |
| Volume-Time Curve |
: |
No |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
No |
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Not Specified |
| Interpretation |
: |
No |
| Software Suite |
: |
Integrates with MasterScope and VIAConnect |
| # of patient records |
: |
1200 |
| HIS Interface |
: |
Not specified |
| BTPS Correction |
: |
Not Specified |
| |
|
|
| Comment |
: |
Intended for clinical trials. Includes Ediary. Sniff nasal PEF optional. Connects with EXPERT and MyStudyPortal. |
| Dimensions |
: |
112x82x37 mm |
| Weight |
: |
167 grams |
| Last Updated / Reviewed |
: |
9/14/2015 |
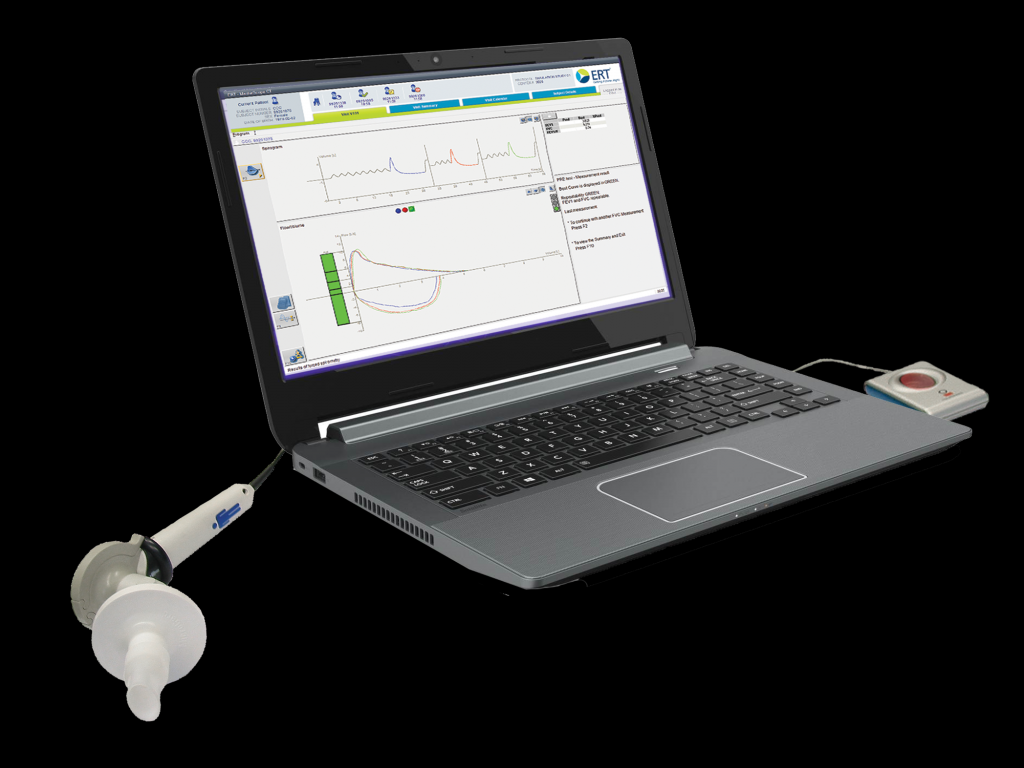 Photo shown with permission of ERT
Photo shown with permission of ERT
| Product Type |
: |
Spirometer |
| Product Name |
: |
Masterscope CT |
| Company Name |
: |
ERT |
| |
|
|
| Intended for |
: |
Portable, Office |
| Configuration |
: |
Hand-held, attached to PC via USB |
| Sensor Technology |
: |
Pneumotach |
| Vol Range & Accuracy |
: |
0.1 – 8.0 L |
| Flow Range & Accuracy |
: |
0.1 to ± 16 L/s, ± 5% of reading or 0.2 L/s |
| Calibration |
: |
Not specified |
| Power Requirements |
: |
Via PC USB port |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
Yes |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
No |
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Not Specified |
| Interpretation |
: |
Not Specified |
| Software Suite |
: |
|
| # of patient records |
: |
Limited only by available disk space |
| HIS Interface |
: |
Not specified |
| BTPS Correction |
: |
Not Specified |
| |
|
|
| Comment |
: |
Intended for clinical trials. ECG included. |
| Dimensions |
: |
Not specified |
| Weight |
: |
Not specified |
| Last Updated / Reviewed |
: |
9/14/2015 |
Pulmonary Function Equipment Buyer's Guide

