| Product Type |
: |
Spirometer |
| Product Name |
: |
Spirolab |
| Company Name |
: |
MIR – Medical International Research |
| |
|
|
| Intended for |
: |
Office |
| Configuration |
: |
Handheld, attached to controller/display unit |
| Sensor Technology |
: |
Turbine |
| Vol Range & Accuracy |
: |
±3% or 50mL |
| Flow Range & Accuracy |
: |
±16L/sec, ±5% o 200mL/s |
| Calibration |
: |
Not required |
| Power Requirements |
: |
Rechargeable battery |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
No |
| MVV |
: |
Yes |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Touch LCD, 7 inch, 800×480 |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
Yes |
| REPORTING: |
|
|
| Printer |
: |
None |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Not Specified |
| Interpretation |
: |
Not Specified |
| Software Suite |
: |
|
| # of patient records |
: |
10,000 records, uploads to PC via bluetooth |
| HIS Interface |
: |
No |
| BTPS Correction |
: |
Yes, built in temperature sensor |
| |
|
|
| Comment |
: |
Oximeter optional. |
| Dimensions |
: |
220 x 210 x 51 mm |
| Weight |
: |
1.45 kg |
| Last updated / reviewed |
: |
01/16/16 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
SmartOne |
| Company Name |
: |
MIR – Medical International Research |
| |
|
|
| Intended for |
: |
Personal |
| Configuration |
: |
Hand-held, connected via bluetooth to Iphone, IPad, Android smartphone or tablet |
| Sensor Technology |
: |
Turbine |
| Vol Range & Accuracy |
: |
±3% or 50mL |
| Flow Range & Accuracy |
: |
±3% or 50mL |
| Calibration |
: |
Not required |
| Power Requirements |
: |
Rechargeable battery |
| TESTS: |
|
|
| FVC |
: |
FEV1 and PEF |
| Pre/Post |
: |
No |
| SVC |
: |
No |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses smartphone or tablet display |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
No |
| Volume-Time Curve |
: |
No |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
No |
| REPORTING: |
|
|
| Printer |
: |
None |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Yes |
| Interpretation |
: |
No |
| Software Suite |
: |
|
| # of patient records |
: |
Limited by available memory |
| HIS Interface |
: |
No |
| BTPS Correction |
: |
No |
| |
|
|
| Comment |
: |
|
| Dimensions |
: |
Not specified |
| Weight |
: |
Not Specified |
| Last updated / reviewed |
: |
01/16/16 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
Minispir Light |
| Company Name |
: |
MIR – Medical International Research |
| |
|
|
| Intended for |
: |
Office |
| Configuration |
: |
Hand-held, downloads to PC via USB |
| Sensor Technology |
: |
Turbine |
| Vol Range & Accuracy |
: |
±3% or 50mL |
| Flow Range & Accuracy |
: |
±16L/sec, ±5% o 200mL/s |
| Calibration |
: |
Not required |
| Power Requirements |
: |
Via PC USB port |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
Yes |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
Yes |
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Not Specified |
| Interpretation |
: |
Yes |
| Software Suite |
: |
WinSpiro |
| # of patient records |
: |
Limited only by available disk space |
| HIS Interface |
: |
HL7 |
| BTPS Correction |
: |
Yes, built in temperature sensor |
| |
|
|
| Comment |
: |
|
| Dimensions |
: |
Not specified |
| Weight |
: |
Not Specified |
| Last updated / reviewed |
: |
01/19/16 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
Minispir New |
| Company Name |
: |
MIR – Medical International Research |
| |
|
|
| Intended for |
: |
Office |
| Configuration |
: |
Hand-held, downloads to PC via USB |
| Sensor Technology |
: |
Turbine |
| Vol Range & Accuracy |
: |
±3% or 50mL |
| Flow Range & Accuracy |
: |
±16L/sec, ±5% o 200mL/s |
| Calibration |
: |
Not required |
| Power Requirements |
: |
Via PC USB port |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
Yes |
| MVV |
: |
Yes |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
Yes |
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Not Specified |
| Interpretation |
: |
Yes |
| Software Suite |
: |
WinSpiro |
| # of patient records |
: |
Limited only by available disk space |
| HIS Interface |
: |
HL7 |
| BTPS Correction |
: |
Yes, built in temperature sensor |
| |
|
|
| Comment |
: |
Oximeter optional. |
| Dimensions |
: |
Not specified |
| Weight |
: |
Not Specified |
| Last updated / reviewed |
: |
01/19/16 |
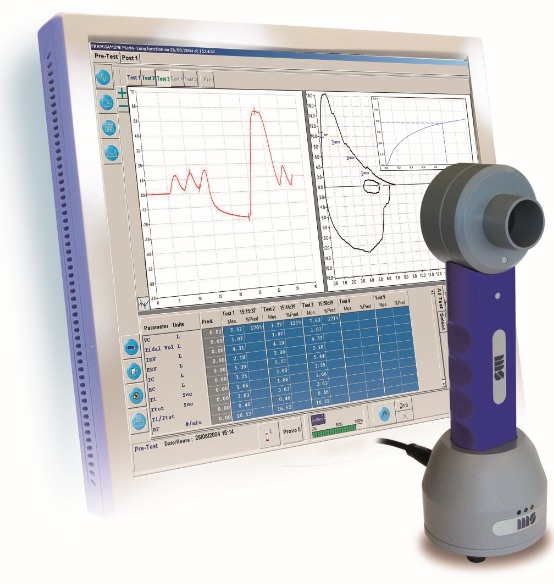 Photo included with permission from Medisoft
Photo included with permission from Medisoft
| Product Type |
: |
Spirometer |
| Product Name |
: |
Micro 6000 |
| Company Name |
: |
Medisoft |
| |
|
|
| Intended for |
: |
Office |
| Configuration |
: |
Hand-held, downloads to PC via Bluetooth |
| Sensor Technology |
: |
Pneumotach |
| Vol Range & Accuracy |
: |
Not specified |
| Flow Range & Accuracy |
: |
0.02 to 15 L/sec, error max < 3% |
| Calibration |
: |
3 Liter syringe |
| Power Requirements |
: |
9V rechargeable battery |
| |
|
|
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
Yes |
| MVV |
: |
Yes |
| Challenge |
: |
Yes |
| |
|
|
| DISPLAY: |
|
|
| Type/Size |
: |
PC display |
| Tidal Loop |
: |
During test and test review |
| Flow-Volume Loop |
: |
During test and test review |
| Volume-Time Curve |
: |
During test and test review |
| Numerical Results |
: |
Test review |
| Comparison |
: |
Test review |
| Incentive |
: |
Not specified |
| |
|
|
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Yes |
| Trends |
: |
Yes |
| Interpretation |
: |
Yes |
| # of patient records |
: |
Limited only by available disk space |
| HIS Interface |
: |
Not specified |
| BTPS Correction |
: |
Not Specified |
| |
|
|
| Comment |
: |
|
| Dimensions |
: |
135 mm x 60 mm |
| Weight |
: |
340 g |
| Last updated / reviewed |
: |
03/09/14 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
SpiroScout |
| Company Name |
: |
Ganshorn Medizin Electronic |
| |
|
|
| Intended for |
: |
Lab |
| Configuration |
: |
Not specified |
| Sensor Technology |
: |
Ultrasonic |
| Vol Range & Accuracy |
: |
Not specified |
| Flow Range & Accuracy |
: |
Not specified |
| Calibration |
: |
Not specified |
| Power Requirements |
: |
Not specified |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
Not Specified |
| MVV |
: |
Not Specified |
| Challenge |
: |
Not Specified |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
Yes |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
Not specified |
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Not Specified |
| Interpretation |
: |
Not Specified |
| Software Suite |
: |
|
| # of patient records |
: |
Not specified |
| HIS Interface |
: |
Not specified |
| BTPS Correction |
: |
Yes |
| |
|
|
| Comment |
: |
Options include Rhinomanometry, Forced Oscillation, Capnovolumetry, Dead Space Measurement |
| Dimensions |
: |
Not Specified |
| Weight |
: |
Not Specified |
| Last updated / reviewed |
: |
9/14/2015 |
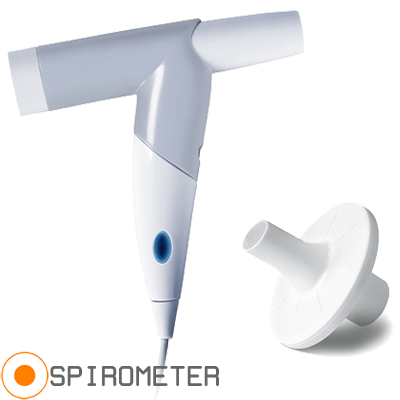 Photo shown with permission of AME
Photo shown with permission of AME
| Product Type |
: |
Spirometer |
| Product Name |
: |
AME Spirometer |
| Company Name |
: |
AME – Advanced Medical Engineering |
| |
|
|
| Intended for |
: |
Portable/Office |
| Configuration |
: |
Hand-held, attached to PC via USB |
| Sensor Technology |
: |
Pneumotach |
| Vol Range & Accuracy |
: |
Not specified |
| Flow Range & Accuracy |
: |
+/- 16 L/sec |
| Calibration |
: |
Not specified |
| Power Requirements |
: |
Via PC USB port |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
Yes |
| SVC |
: |
Yes |
| MVV |
: |
Yes |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
Yes |
| Pediatric Incentive |
: |
Yes |
| REPORTING: |
|
|
| Printer |
: |
External, attached to PC |
| User Configurable Reports |
: |
Not Specified |
| Trends |
: |
Yes |
| Interpretation |
: |
No |
| Software Suite |
: |
|
| # of patient records |
: |
Not specified |
| HIS Interface |
: |
No |
| BTPS Correction |
: |
Not Specified |
| |
|
|
| Comment |
: |
Tilt sensors: left, right, forward, back. Can email jpeg of results. Can be used with AME weather station. |
| Dimensions |
: |
14 x 15 x 0.45 cm |
| Weight |
: |
200 g |
| Last Updated |
: |
9/8/2015 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
SM-1 |
| Company Name |
: |
Nickel-Electro Ltd. |
| |
|
|
| Intended for |
: |
Student |
| Configuration |
: |
Table-top |
| Sensor Technology |
: |
Volume Displacement |
| Vol Range & Accuracy |
: |
0-7 L |
| Flow Range & Accuracy |
: |
Not specified |
| Calibration |
: |
Not required |
| Power Requirements |
: |
Not required |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
No |
| SVC |
: |
No |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
None |
| Type/Size |
: |
No |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
Yes, with data logger |
| Volume-Time Curve |
: |
No |
| Numerical Results |
: |
No |
| Comparison |
: |
No |
| Pediatric Incentive |
: |
None |
| REPORTING: |
|
|
| Printer |
: |
No |
| User Configurable Reports |
: |
No |
| Trends |
: |
No |
| Interpretation |
: |
Requires data logger to record results |
| Software Suite |
: |
None |
| # of patient records |
: |
No |
| HIS Interface |
: |
No |
| BTPS Correction |
: |
No |
| |
|
|
| Comment |
: |
|
| Dimensions |
: |
720x320x230 mm |
| Weight |
: |
Not Specified |
| Last Updated |
: |
1/20/2016 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
Pasport Spirometer |
| Company Name |
: |
PASCO |
| |
|
|
| Intended for |
: |
Student |
| Configuration |
: |
Hand-held, attached base unit and then to PC via USB |
| Sensor Technology |
: |
Pneumotach |
| Vol Range & Accuracy |
: |
Not specified |
| Flow Range & Accuracy |
: |
Not specified |
| Calibration |
: |
Not specified |
| Power Requirements |
: |
Via base unit |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
No |
| SVC |
: |
No |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
No |
| Flow-Volume Loop |
: |
No |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
No |
| Comparison |
: |
No |
| Pediatric Incentive |
: |
No |
| REPORTING: |
|
|
| Printer |
: |
None |
| User Configurable Reports |
: |
No |
| Trends |
: |
No |
| Interpretation |
: |
No |
| Software Suite |
: |
Requires PASPORT Interface |
| # of patient records |
: |
None |
| HIS Interface |
: |
No |
| BTPS Correction |
: |
No |
| |
|
|
| Comment |
: |
|
| Dimensions |
: |
Not Specified |
| Weight |
: |
Not Specified |
| Last Updated |
: |
1/20/2016 |
| Product Type |
: |
Spirometer |
| Product Name |
: |
Human Respiratory Kit |
| Company Name |
: |
AD Instruments |
| |
|
|
| Intended for |
: |
Student |
| Configuration |
: |
Hand-held, attached base unit and then to PC via USB |
| Sensor Technology |
: |
Pneumotach |
| Vol Range & Accuracy |
: |
Not specified |
| Flow Range & Accuracy |
: |
+/- 1000 LPM |
| Calibration |
: |
Not specified |
| Power Requirements |
: |
Via PC USB port |
| TESTS: |
|
|
| FVC |
: |
Yes |
| Pre/Post |
: |
No |
| SVC |
: |
No |
| MVV |
: |
No |
| Challenge |
: |
No |
| DISPLAY: |
|
|
| Type/Size |
: |
Uses PC display |
| Tidal Loop |
: |
Yes |
| Flow-Volume Loop |
: |
Yes |
| Volume-Time Curve |
: |
Yes |
| Numerical Results |
: |
Yes |
| Comparison |
: |
No |
| Pediatric Incentive |
: |
No |
| REPORTING: |
|
|
| Printer |
: |
None |
| User Configurable Reports |
: |
No |
| Trends |
: |
No |
| Interpretation |
: |
No |
| Software Suite |
: |
Requires AD Instruments Spirometry Software |
| # of patient records |
: |
None |
| HIS Interface |
: |
No |
| BTPS Correction |
: |
No |
| |
|
|
| Comment |
: |
|
| Dimensions |
: |
Not Specified |
| Weight |
: |
Not Specified |
| Last Updated |
: |
1/16/2015 |
Pulmonary Function Equipment Buyer's Guide

