The ATS has released its first standard for reporting pulmonary function results. This report is in the December 1, 2017 issue of the American Journal of Respiratory and Critical Care Medicine. At the present time however, despite its importance it is not an open access article and you must either be a member of the ATS or pay a fee ($25) in order to access it. Hopefully, it will soon be included with the other open access ATS/ERS standards.
There are a number of interesting recommendations made in the standard that supersede or refine recommendations made in prior ATS/ERS standards, or are otherwise presented for the first time. Specific recommendations include (although not necessarily in the order they were discussed within the standard):
- The lower limit of normal, where available, should be reported for all test results.
- The Z-score, where available, should be reported for all test results. A linear graphical display for this is recommended for spirometry and DLCO results.
- Results should be reported in tables, with individual results in rows. The result’s numerical value, LLN, Z-score and percent predicted are reported in columns, in that recommended order. Reporting the predicted value is discouraged.
Part of Figure 1 from page 1466 of the ATS Recommendations for a Standardized Pulmonary Function Report.
- All tests should be reported as separate sections. Spirometry, slow vital capacity, lung volumes and DLCO should be reported in that order, with other tests following.
- The reference source for normal values should be included with each section of the report.
- Although not specifically dis-allowed, the use of bold face or colored fonts when displaying results below the LLN is discouraged.
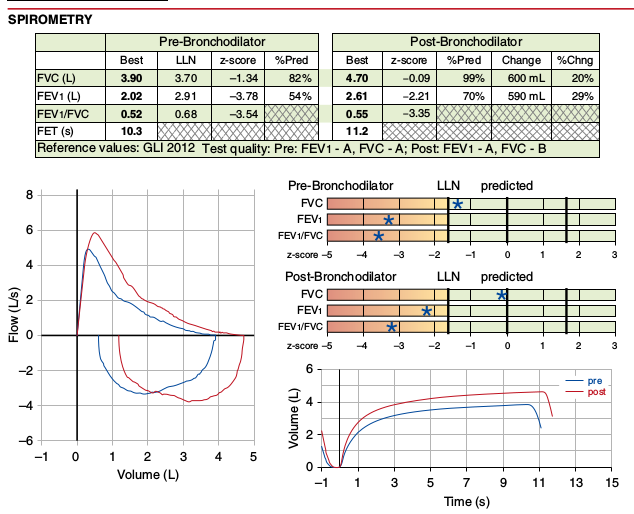
- Only FVC, FEV1 and the FEV1/FVC ratio should be routinely reported for spirometry. Reporting FEF25-75 and instantaneous flows (FEF75% for example) is discouraged. Expiratory time (FET) should be reported for quality assessment purposes.
- The FEV1/FVC ratio should only be reported as a decimal fraction with the LLN and Z-score but its percent of the predicted is specifically discouraged from being reported.
- The use of the GLI spirometry reference equations is recommended, although the NHANES III spirometry reference equations are considered acceptable if continuity is important. No recommendations were made for lung volume or DLCO reference equations.
- Spirometry test quality should be graded using an A-F scale. Although it is recommended that results with a quality score of F should not be reported the standard notes that acceptable test quality is not possible in some subjects and that is up to the interpreting reviewer to decide whether poor quality results are adequate for interpretation or not.
- For spirometry both the flow-volume loop and volume-time curve should be included. Peak flow should be read from the flow-volume loop graph and not reported in the table of spirometry results.
- The size of the flow-volume loop and volume-time curve are mandated. Flow-volume loops are to be no smaller than 5 mm per L/sec and 10 mm per L. Volume-time curves are to be no smaller than 10 mm per L and 20 mm per second. These specifications do not appear to include any numerical scale.
Note: For my lab’s reports a flow-volume loop would be at least 16 cm x 8 cm (+/- 16 L/sec x 8 L) and a volume-time curve would be at least 8 cm x 32 cm (8 L x -1/+15 sec). This is a bit problematic since 32 cm is 12.6 inches and at this scale a volume-time curve would not fit a standard sheet of paper and we’d have to use a smaller length of time.
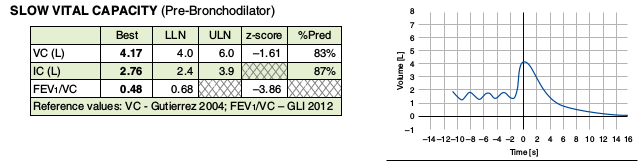
- The SVC should be reported in a separate section and, if available, should include the FEV1/VC ratio. Although an example in the standard included IC as a reported value, no recommendations concerning the reporting of IC and ERV were made within the text of the report.
- The volume-time curve for the SVC should be reported with enough of the volume-time curve to show the end-exhalation baseline and the SVC maneuver.
Part of Figure 1 from page 1466 of the ATS Recommendations for a Standardized Pulmonary Function Report.
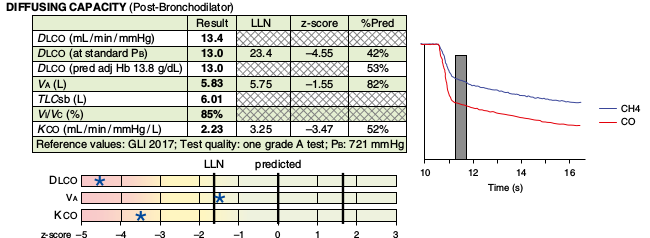
- Reporting KCO from the DLCO is optional but reporting it as DL/VA is specifically discouraged. (Interestingly, the example included in the standard notes KCO to be reported as ml/min/mmHg/L which is essentially incorrect since there is no volume, i.e. ml or L, in KCO which to me indicates a continued and pervasive lack of understanding of the nature of KCO).
- It is recommended that DLCO and DLCO corrected for barometric pressure be reported as separate values. An additional row that takes into consideration the predicted DLCO adjusted for hemoglobin is also recommended.
- Although reporting the DLCO’s VA, TLCsb and Vi/Vc were included within an example within the standard, these results were not specifically addressed.
- Both date of birth and age should be reported. Age should include a decimal point for children and adolescents.
- Barometric pressure should be reported (units are not specified but mm Hg were implied).
- Oxygen saturation is suggested as a demographic item (although how and why is left open to question).
- Whether or not reference values are adjusted for ethnicity or specific to an ethnicity must be included, but this is specified to be in the technician notes, not the tabular results. Interestingly, ethnicity/race is not included among the recommended demographic items.
There are however, a number of reporting and interpretation problems that were not addressed or only partially addressed, and their solutions, if any, left to individual labs and manufacturers.
- It’s possible for multiple post-baseline spirometry tests to be performed such as post-BD, post-exercise and supine, but no easy way to report more than one pair in any report.
- The reported FVC and FEV1 can be taken from different tests but there is no suggested way to indicate that this has occurred. In addition, only one flow-volume loop and volume-time curve can be reported and there are no suggestions whether either or both should be linked to the FEV1 or to the FVC.
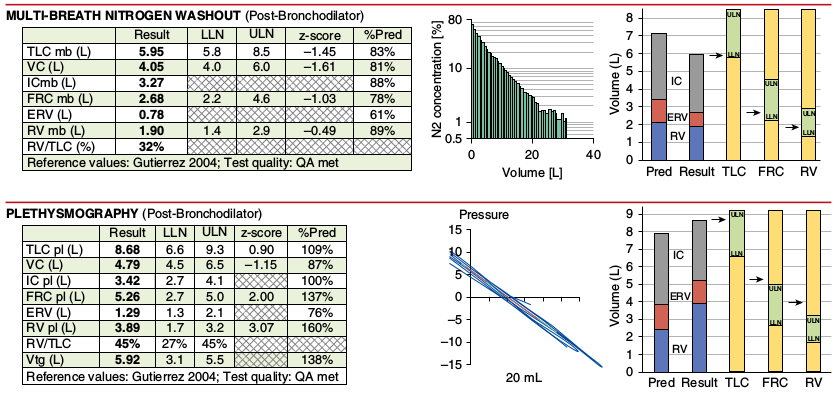
- Multiple N2 washout and plethysmographic lung volume tests can, and often are, performed and their averaged results reported, but graphs can only be taken from a single test and there were no recommendations as to how this single graph is selected.
- It appears that to be specifically recommended that that the helium curve for helium dilution lung volumes not be added to the report. Why this information is considered unimportant while the N2 washout curve and plethysmographic pressure-volume tracing were was not explained.
- It was noted that spirometry efforts with low peak flows can have a higher FEV1 due to reduced negative effort dependence. Although the standard recommends that a technician should coach for maximal effort there is no explicit recommendation that peak flow be used when selecting spirometry efforts.
- The quality grading system for spirometry is based solely on the reproducibility of acceptable tests (both the number of tests and their closeness) but at the same time that standard notes that acceptability (back-extrapolation, expiratory time, EOT flow rate) needs to be assessed based on the limitations or abilities of the subject. However, almost none of these acceptability factors (other than what can be determined from flow-volume loops and volume-time curves) are specifically recommended to be reported and in fact, reporting of them appears to be discouraged.
- A graph showing the washout phase of the DLCO test with the alveolar sample was shown and suggested for inclusion in reports however this graph does not show exhaled volume and the 2017 DLCO standard recommends that graphs of both the “full manoeuvre and exhaled gas concentration versus volume with sample collection” be reported.
- Although additional tests such as Oscillometry, MIP/MEP and Exhaled NO were mentioned in passing (and I’ll add 6MWT and HAST testing), other than giving very general guidelines, there were no examples and no particular suggestions about reporting additional tests.
The ATS standards on reporting are welcome. I’ve seen reports from many different PFT labs and the lack of standardization is notable. In addition, many reports are missing important information that the new ATS reporting standard more or less mandates. Having said all this, the standard is a first effort and there are a number of issues, some of which I consider to be relatively basic, that were not addressed. In addition, a careful reading of the reporting standard shows that although some factors are mandated, many are not and much of the decision about what should and should not be included in a report is still up to individual labs and manufacturers. Although there were some specific recommendations there were no overall guidelines for which results must be reported; which results are optional; and which results must be excluded for spirometry, lung volumes and DLCO.
A large part of the standard was devoted to quality assessment, but there were some inconsistencies, the most notable of which was the lack of any quality assessment for lung volume measurements. Moreover, much of what was said about quality assessment is to one extent or another derived from previous ATS/ERS standards and not original. Relatively strict grading systems for spirometry and diffusing capacity were recommended but the limitations of these systems were also extensively noted and this leaves their inherent value open to question. My criticism of grading systems like these is that they are focused on factors that relatively easy to measure but leave the more difficult factors to the technician or interpreting physician. Even so, what’s important is that the standard does mandate the reporting of test quality.
One very important issue that should have been included was standardized test nomenclature. This is implied to some extent in the included examples, but there are discrepancies particularly in the use of acronyms, capitalization, subscripts and subscripts between this standard and prior ATS/ERS standards. Because there is no recommended nomenclature this means that individual labs will continue to use the nomenclature they are comfortable with.
Another issue that should have been discussed was trend reporting. As important as current test results may be, comparing them over time is at least equally important. Trend reports should be mandated and not left to the discretion of individual PFT labs and manufacturers.
Finally, although not directly related to the reporting standard, equipment manufacturers need to develop friendlier and more responsive report management software. My lab’s reporting software has remained essentially unchanged for at least the last 15 years. Creating, modifying, selecting and printing reports continues to be a time-intensive process with much of the “heavy lifting” done by us and not the software.
In addition there is a reporting issue that appears to be more or less universal and that is the use of fixed-format pages where the positions of all the report elements are decided in advance. I think we can all agree that in order to make reports readable tests that aren’t performed (i.e. empty tables and graphs) shouldn’t be on the report. But since all report elements are fixed in advance this means that my lab has to maintain over a half dozen different report formats for our most common combinations of tests. Even so, we still perform unusual combinations of tests on occasion and when this happens our only recourse (other than creating a new report format on the spot) is to use our “kitchen sink” report that includes all of the tests we are able to perform and is 5 pages long. As importantly, any changes made to a single report often have to be manually duplicated the other half dozen report formats.
Over 20 years ago, the DOS version of our lab software (during the pre-Windows stone age for all the youngsters out there) allowed us to format the individual sections of a report and when printed, only the sections that had test results were printed, This meant there was really only one report format no matter what combination of tests we performed but here we are 20 years later with far more advanced computers and software and we’re stuck having to manually maintain and select a bunch of different report formats. Not exactly what I’d call progress.
References:
Culver BH, Graham BL, Coates AL, et al. Recommendations for a standardized pulmonary function reports. Am J Respir Crit Care Med 2017; 196(11): 1463-1472
Graham BL, Brusasco V, Burgos F, et al. 2017 ERS/ATS standard for single-breath carbon monoxide uptake in the lung. Eur Respir J 2017; 49: 1600016.

PFT Blog by Richard Johnston is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License

Thanks Richard! This is great info.
Anita –
Thanks. I think that most of us however, are going to have to wait until our software is updated to be able to implement the most important recommendations. Given that my equipment’s manufacturer has been exceedingly slow with any software updates I’m not holding my breath.
Regards, Richard
Reading from the UK and another great article thanks to keep me up to date on things. Also will be waiting on the manufacturers to catch up in the NHS era but to also have consultants accept no Predicted value will be a challenge as ours didn’t like the LLN or ULN
Will –
As I mentioned we’re still waiting five years after they were published to be able to use the GLI spirometry reference equations, so at this point I wouldn’t suggest that anybody hold their breath.
Everybody who’s anybody in Pulmonary Medicine seems to have jumped onto the LLN as the be-all and end-all approach for interpreting PFTs. I understand this but I also feel that it’s just another line in the sand and in it’s own way, just as arbitrary as using 80% of predicted as a cutoff. Really, what’s magic about the bottom 5% of a given study population being considered abnormal? Why not 6% or 4%? The problem is that there is no easy way to define normalcy in pulmonary function results and you’ve got to make the best choice you can. Despite some of my reservations about the LLN however, I do think that the Z-score is a better way to think about PFT results than the percent of predicted. The example I use when talking about this is that for somebody with severe COPD, an increase in FEV1 from 0.50 L to 0.75 L makes a profound difference in their clinical status and a reasonably large change in Z-score but is only a couple percent of predicted. Using percent predicted implies that there is a linear gradient in PFT results vs clinical status and this just isn’t true.
Warm regards, Richard
Thanks for the information – most useful. We are in the process of designing our own database for PFT results. Vendor supplied reporting systems are so limited in application. A custom database gives much more flexibility and allows for use of GLI equations. But it takes a lot of work (18 months so far) and coding skills to make it happen. All of our physicians accept the use of LLN – time to introduce Z scores into the mix!
Hi Richard,
Thanks for the article – really interesting and happy to know other labs share our fustration with report templates. We are currently undergoing an upgrade to a new software which I am sure you have heard of (SentrySuite) which gives you the ability to have one report template for all tests and will not display a table for testing that has not been done (I hope that has made sense!). We have all our RFTs on one page (Spirometry, DLCO, Lung Volumes, MIPS/MEPS, FeNO and ABGs and it will only populate the tables with data). We have a separate page for 6MWT and bronchoprovocation testing.
We also find a lot of the information that ATS/ERS suggest are mainly geared towards other scientists and not very clincally significant. I believe most respiratory physicians/registrars will not know how the ‘pressure-volume’ graph of a body pleth manoeuvre is suppose to look like. I think these graphs are great for the head scientist or whoever is responsible for ensuring results are accurate and reproducible, but providing them on a report i feel can make the whole report messy and more complicated then it actually is.
Regards, Oliver
Oliver –
The point of the graphs is to assist the reviewing physician in assessing test quality. Their ability to do this is limited by the fact that the reported values for DLCO and LV tests are usually averages of multiple tests but the graphs come from a single test which may or may not be representative. In addition, it seems that the details of pulmonary function testing are only rarely taught to physicians nowadays (and I include pulmonologists in this) so the information about test quality contained in the graphs often goes unseen. I guess this means that I agree with you that graphs of DLCO and LV tests make a report more complicated than it should be.
Regards, Richard